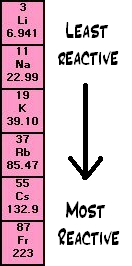
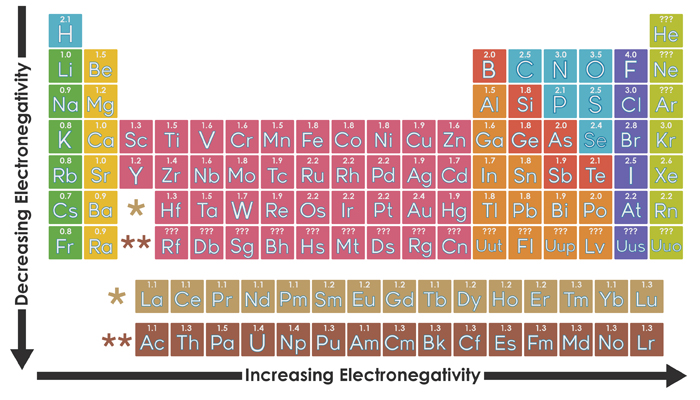
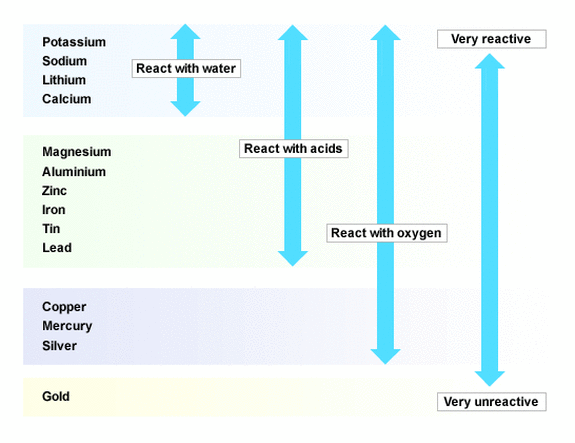
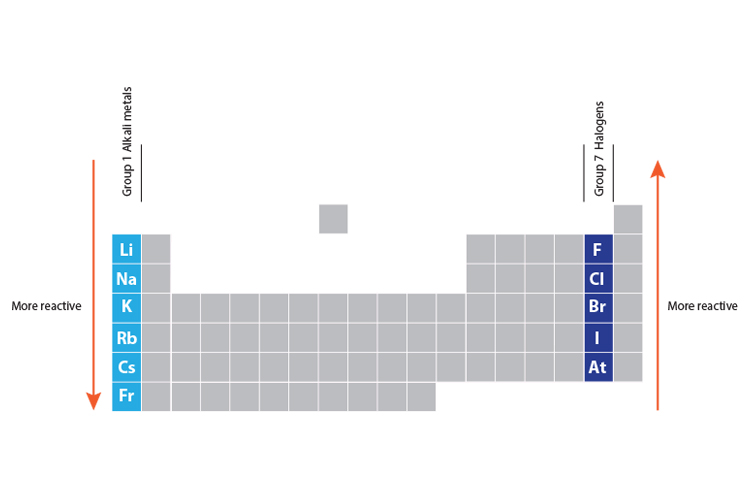
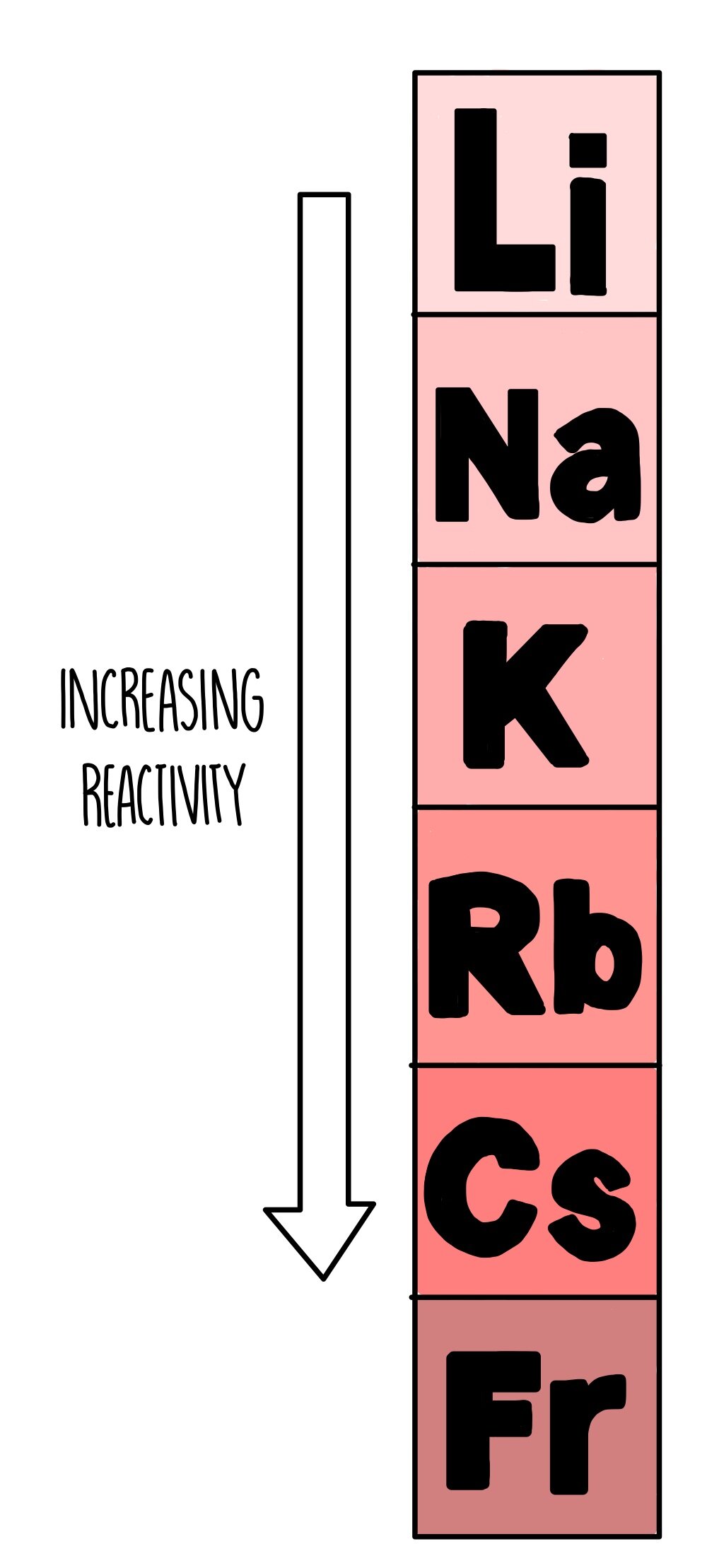
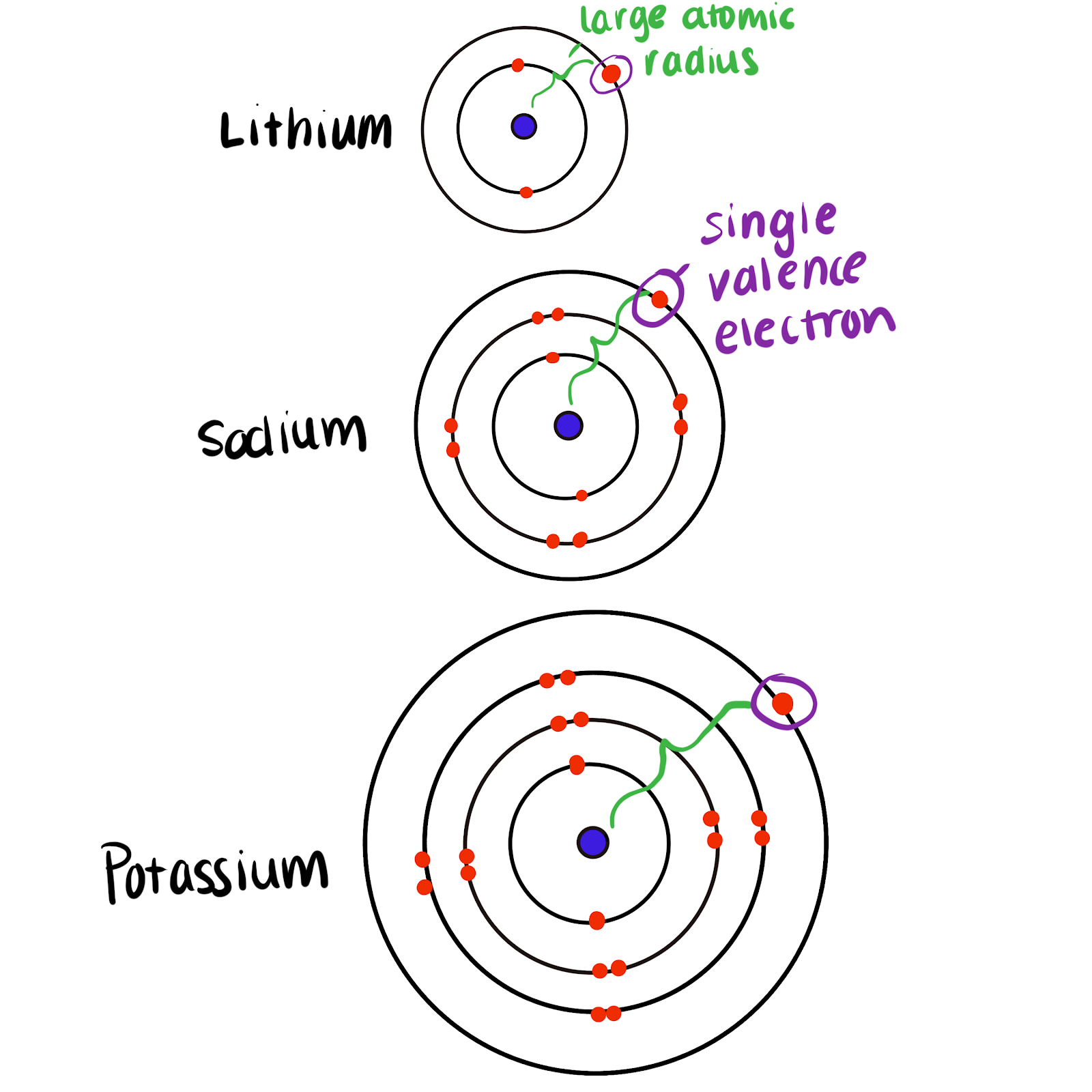
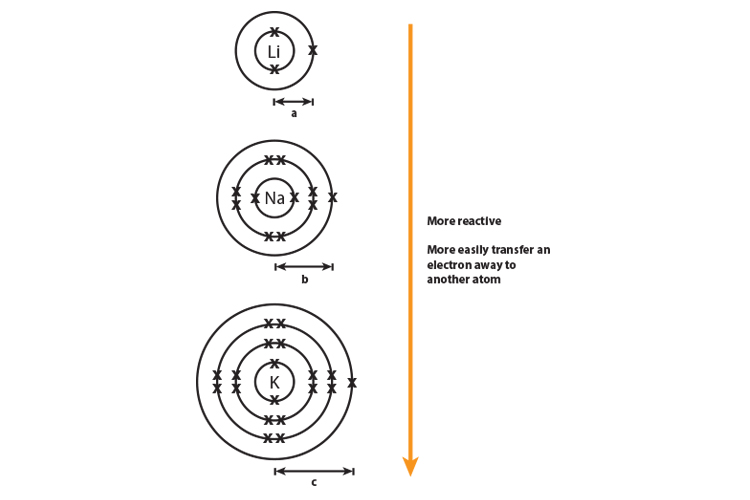
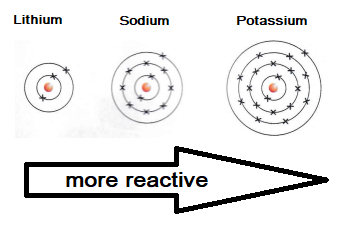
gcse Group 1 ALKALI METALS lithium sodium potassium physical properties balanced equations explaining reactivity trend chemical reactions uses KS4 science igcse O Level chemistry revision notes

Reactivity of alkali and alkaline earth metal tetrafluorobromates towards aromatic compounds and pyridine - ScienceDirect

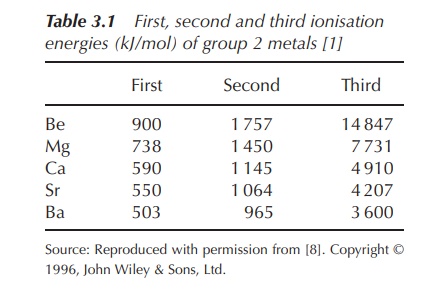
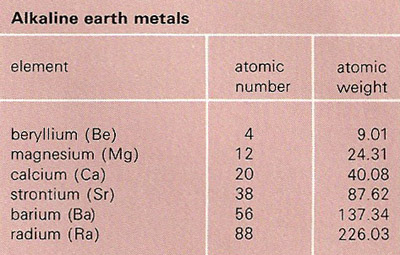
Group 2: Alkaline Earth Metals (Be, Mg, Ca, Sr, Ba, Ra ) Most are metals that react with water to give X(OH) 2 and with oxygen to give XO. Reactivity increases. - ppt download
Towards designing reactive glasses for alkali activation: Understanding the origins of alkaline reactivity of Na-Mg aluminosilicate glasses | PLOS ONE

Comparing halogen reactivity trends with those of the alkali metals. Halogens become less reactive down t… | Chemistry lessons, Study chemistry, Chemistry classroom