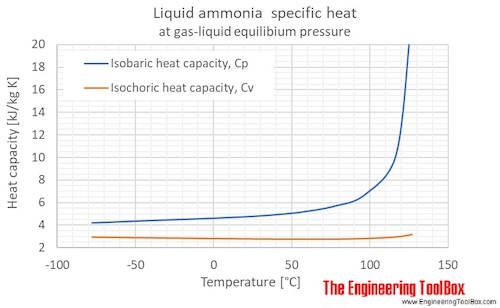
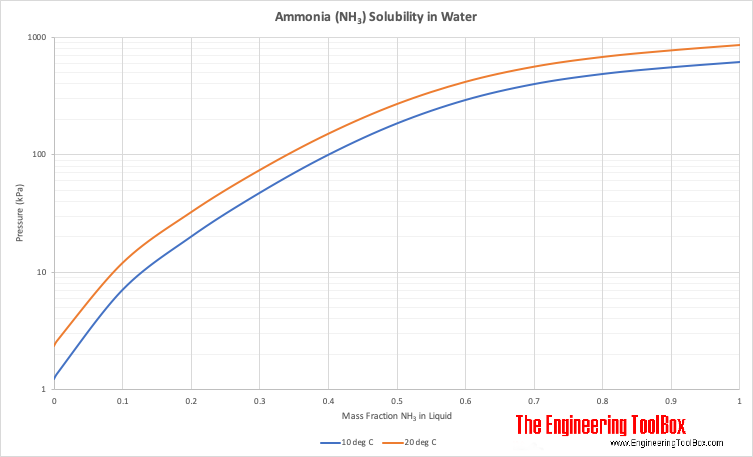
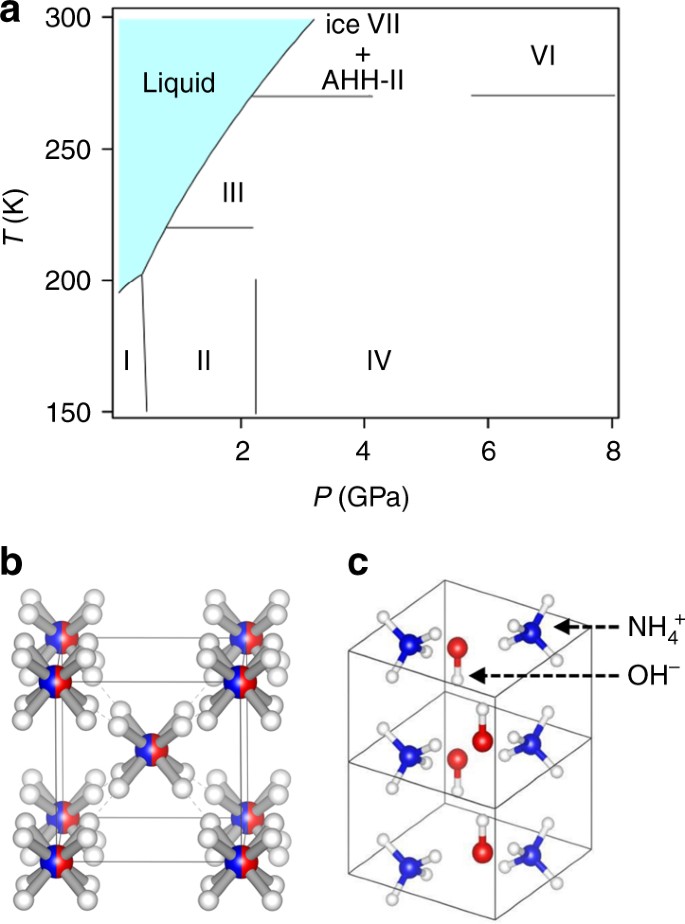
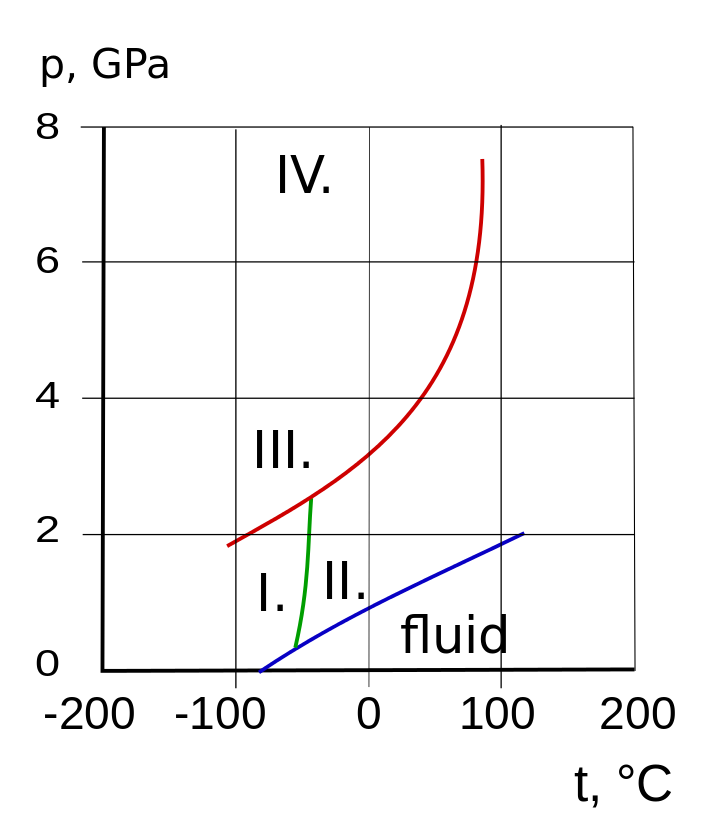
The Liquidus Temperature for Methanol‐Water Mixtures at High Pressure and Low Temperature, With Application to Titan - Dougherty - 2018 - Journal of Geophysical Research: Planets - Wiley Online Library

If the freezing point of a `0.01` molal aqueous solution of a cobalt (III) chloride-ammonia - YouTube

The Liquidus Temperature for Methanol‐Water Mixtures at High Pressure and Low Temperature, With Application to Titan - Dougherty - 2018 - Journal of Geophysical Research: Planets - Wiley Online Library

If the freezing point of a 0.01 molal aqueous solution of a cobalt (III) chloride-ammonia complex (which behaves as a strong electrolyte) is -0.0558^(@)C, the number of chloride (s) in the coordination



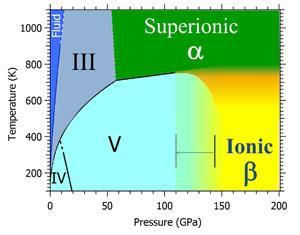

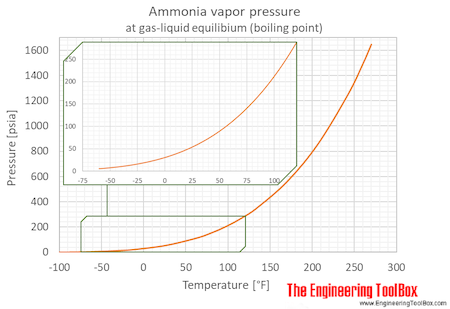
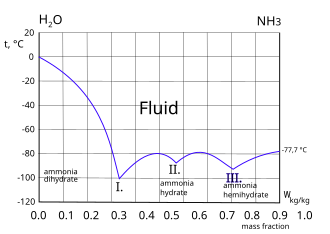
![ammonia-water phase equilibrium diagram [18]. | Download Scientific Diagram ammonia-water phase equilibrium diagram [18]. | Download Scientific Diagram](https://www.researchgate.net/publication/263845643/figure/fig2/AS:669532045705228@1536640287287/ammonia-water-phase-equilibrium-diagram-18.png)