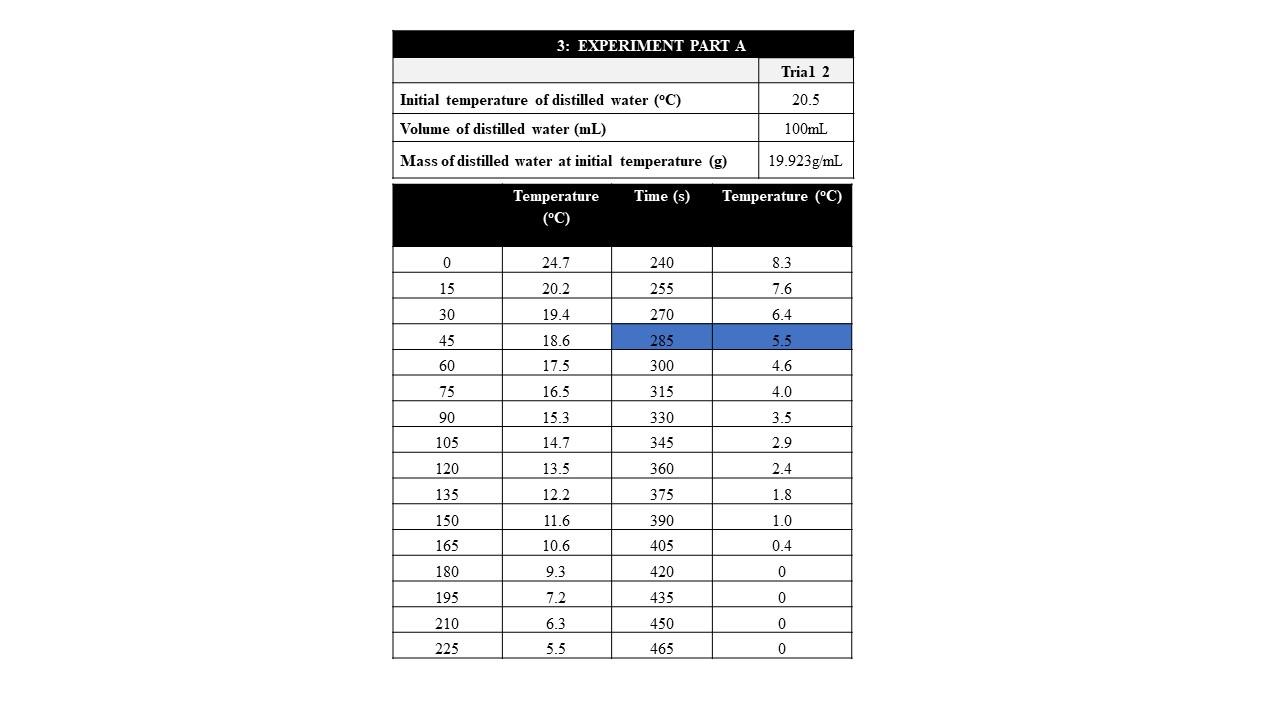
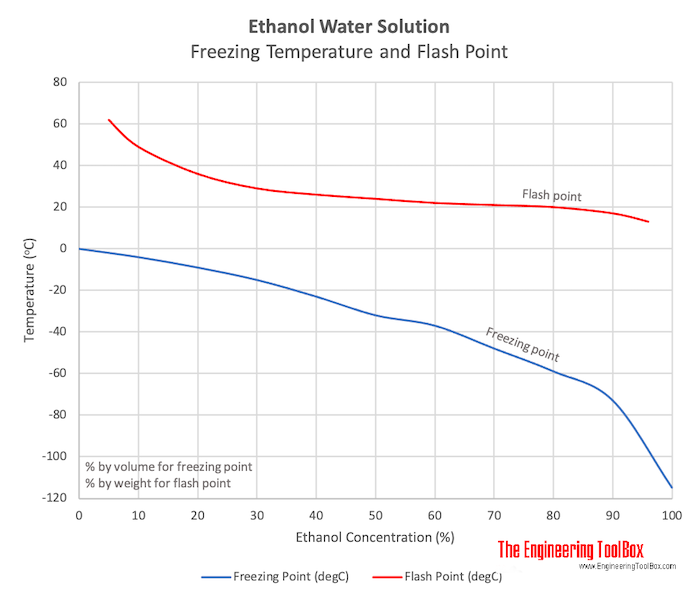
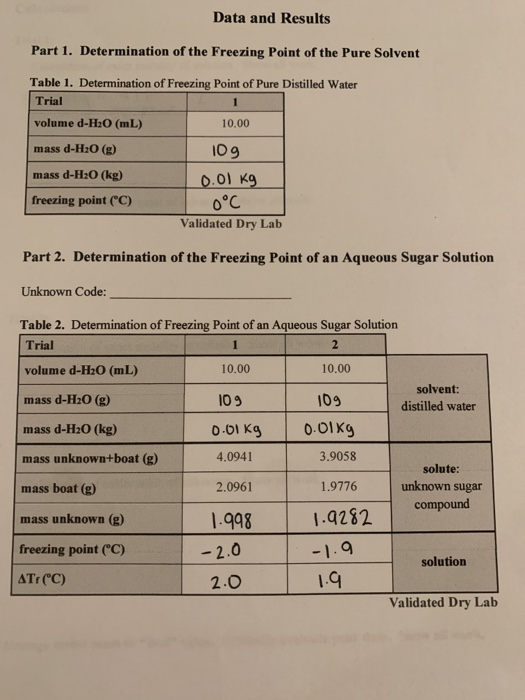
YukonU Youth Moving Mountains - Explore how different substances affect water's freezing point with this fun ice melting experiment! 🧊 Ice forms when water freezes. Pure distilled water has a melting/freezing point

A solution of CaCl(2) was prepared by dissolving 0.0112g in 1 kg of distilled water (molar mass of Ca = 41 g mol^(-1) and Cl =35.5 g mol^(-1)). The freezing point constant

SOLVED: 6. How would the freezing and boiling points of distilled water and seawater compare? Both the freezing and boiling - points of seawater would be lower: Both the freezing and boiling