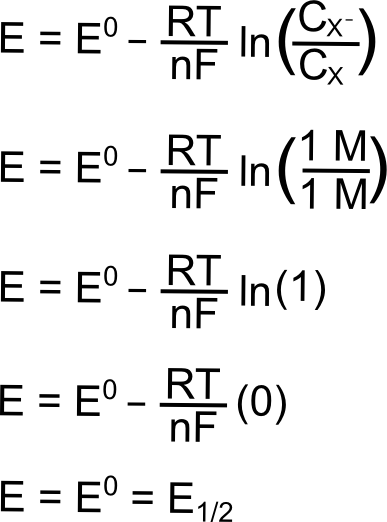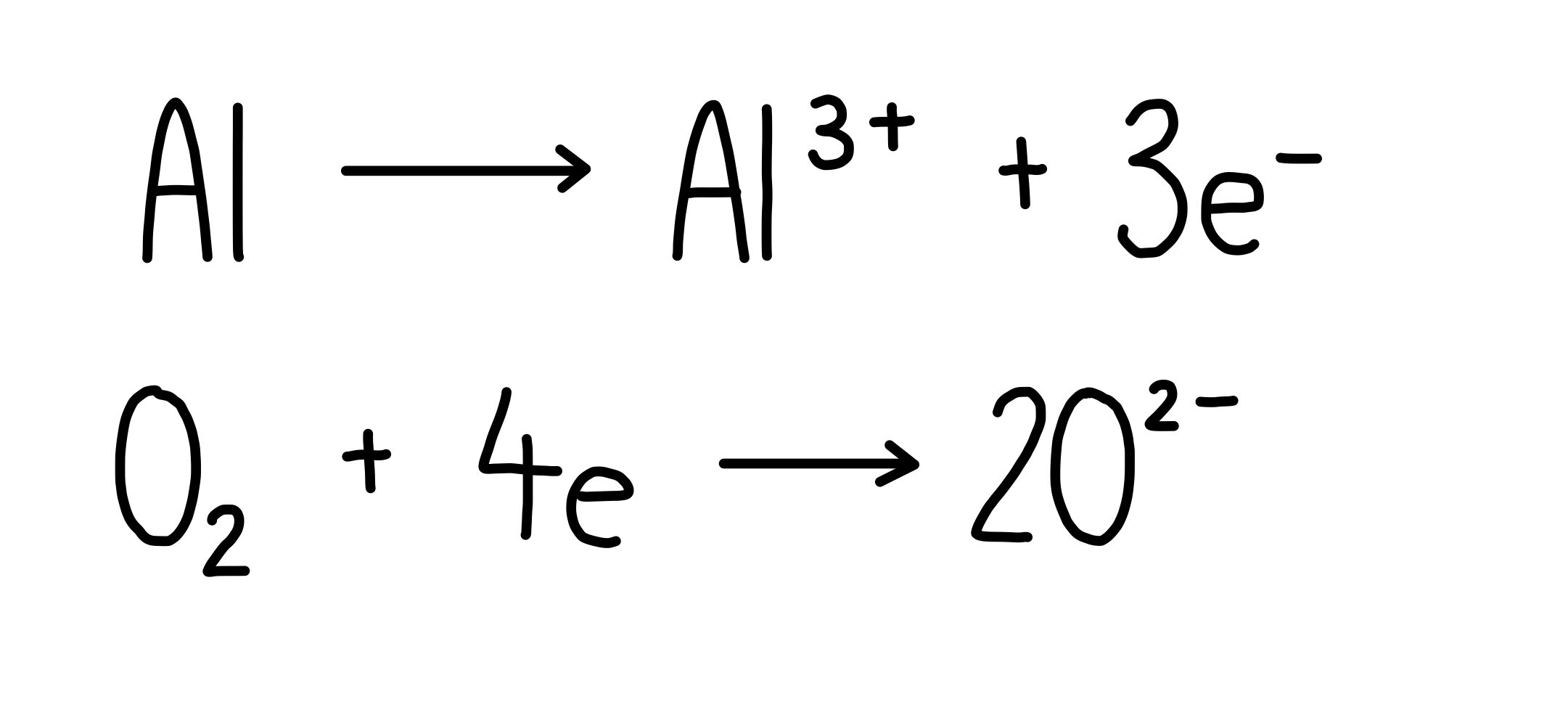How to find the electrode potentials of hydrogen electrodes at 25 °C and 100 kPa in solutions of pH 0 and pH 14 - Quora

Electrochemistry #9| Nernst Equation | Find Electrode Potential or EMF at any concentration - YouTube

during finding half cell electrode potential by nernst equation whether cofficent is used as raise to power or not lr9gj5oo -Chemistry - TopperLearning.com
What is single electrode potential ? How would you calculate the single electrode potential ? - Sarthaks eConnect | Largest Online Education Community

equilibrium - Calculate the cathode electrode potential in this redox reaction - Chemistry Stack Exchange

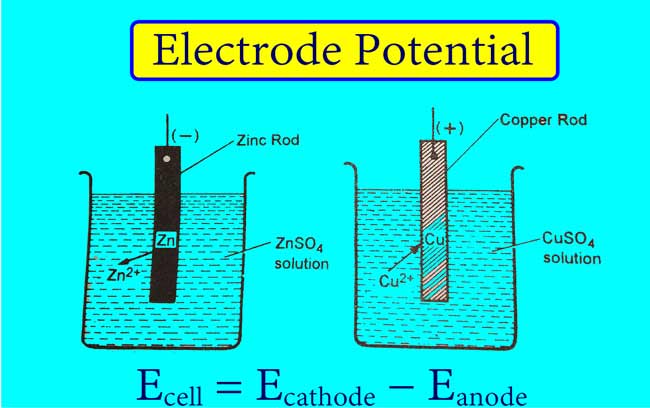
1 Calculating the Cell Potential The process of calculating the cell potential is simple and involves calculation of the potential of each electrode separately, - ppt download
Electric Potential Energy: Definition, Formula and Example | Electrode and Electrode Potential – Chemistry Page

Calculate the standard electrode potential of the Ni^(2+)//Ni electrode , if the cell potential ... - YouTube

Half-cell potentials Electrochemical Series using E cell predicting reaction feasibility A level GCE AS A2 chemistry revision notes KS5