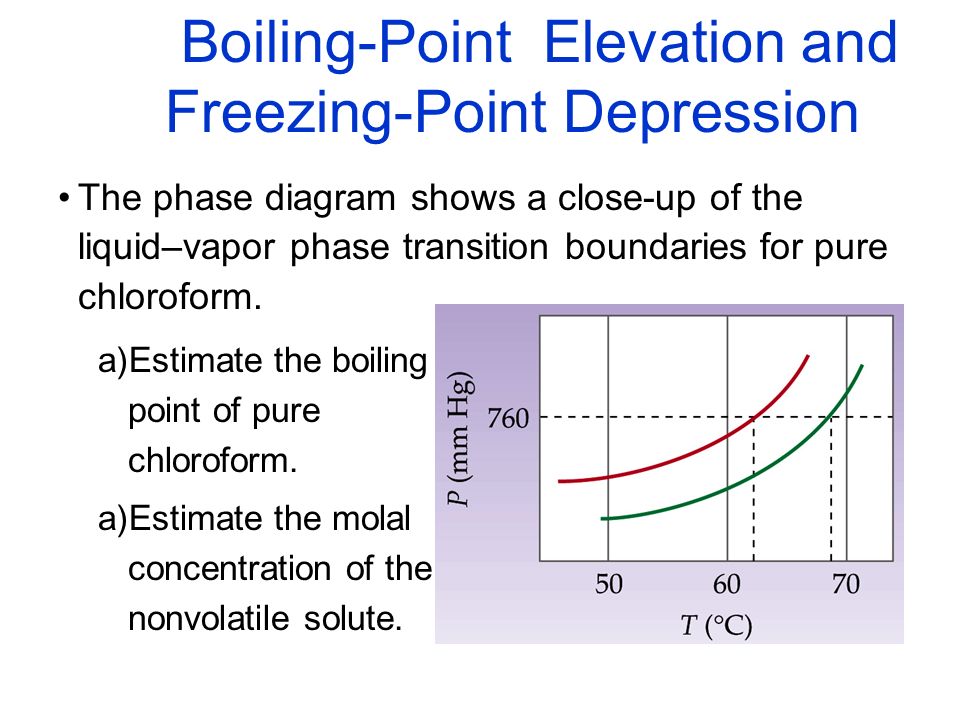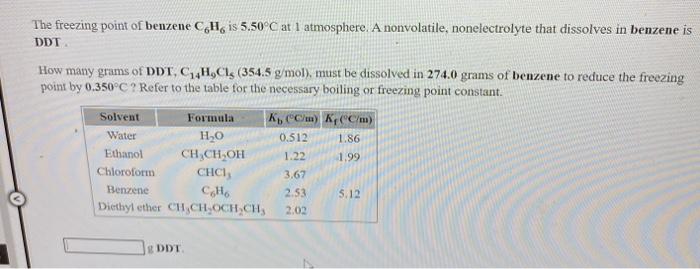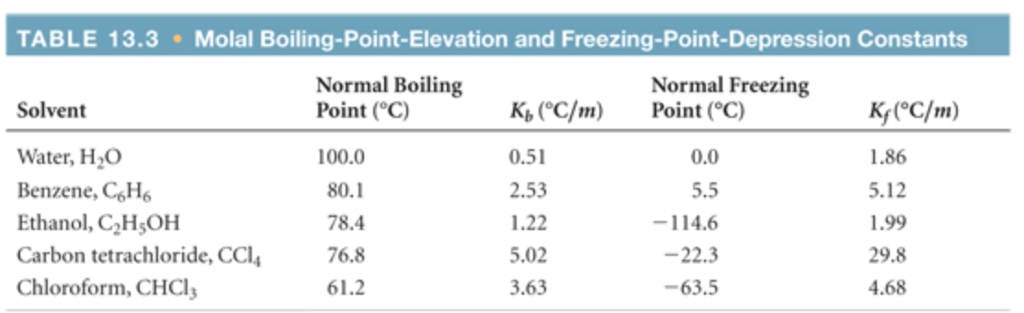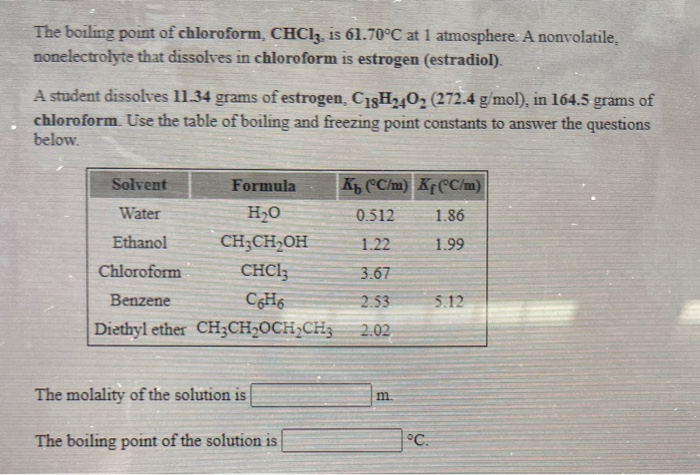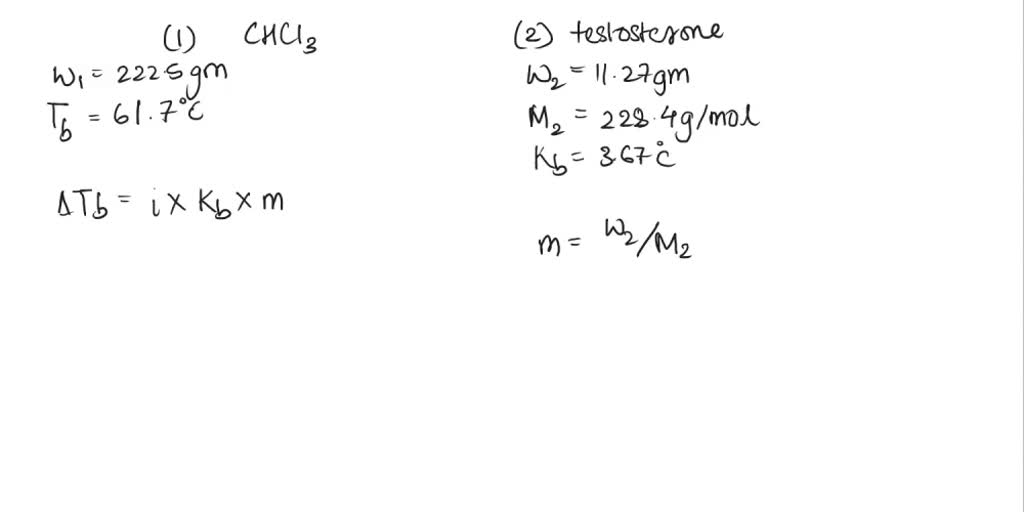
SOLVED: The boiling point of chloroform, CHCl3, is 61.70°C at 1 atmosphere. A nonvolatile, nonelectrolyte that dissolves in chloroform is testosterone. A student dissolves 11.27 grams of testosterone, C19H28O2 (288.4 g/mol), in

Check: What is the molality of a solution of naphthalene dissolved in chloroform if the solution has a - Brainly.com

SOLVED: The freezing point of benzene C6H is 5.50-C at 1 atmosphere nonvolatile; nonelectrolyte that dissolves in benzene is TNT (trinitrotoluene). How many grams of TNT; C H;N3O6 (227.1 g mol): must

SOLVED: The freezing point of pure chloroform is -63.5°C, and its freezing point depression constant is 4.07°C•kg/mol. If the freezing point of a solution of benzoic acid in chloroform is -70.55°C, what