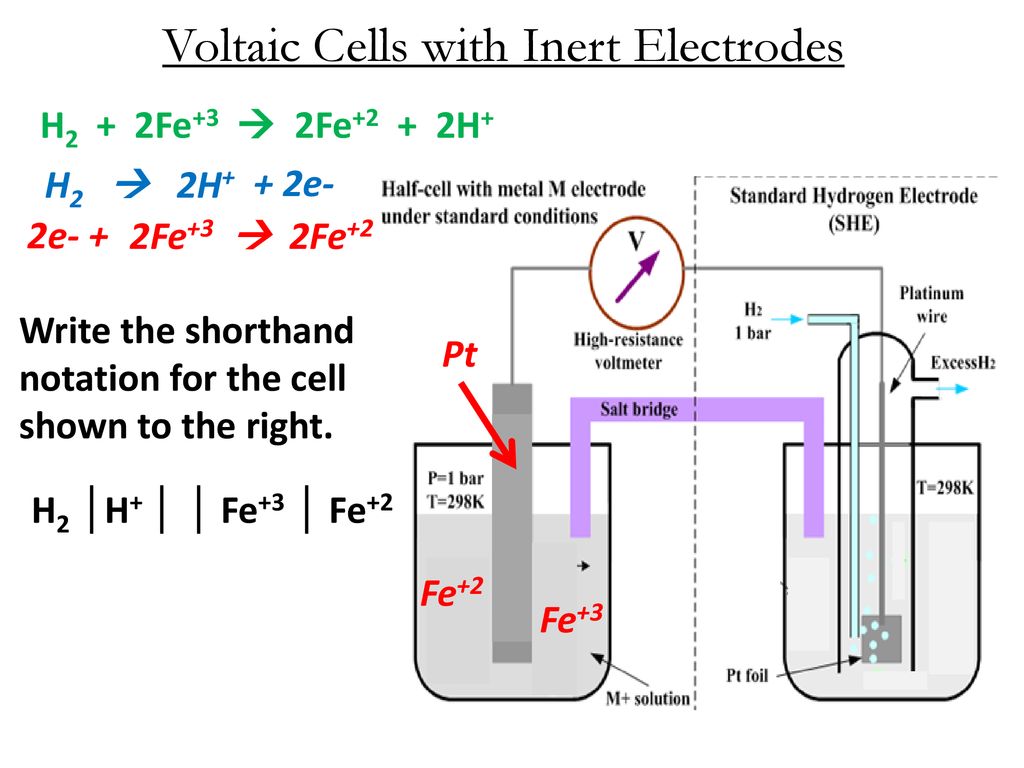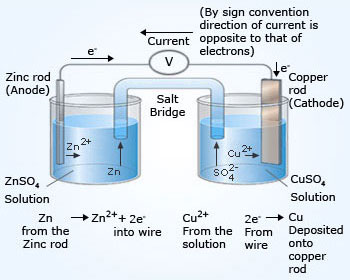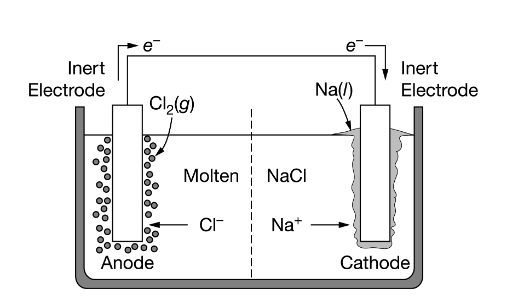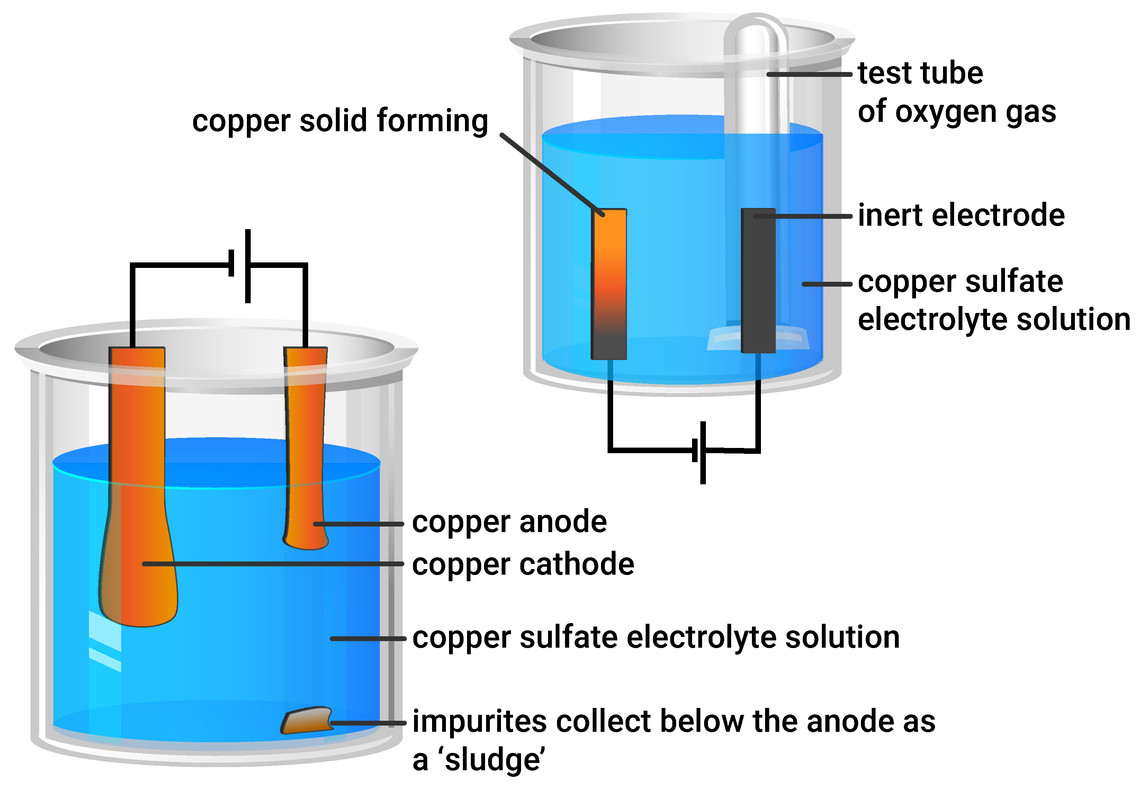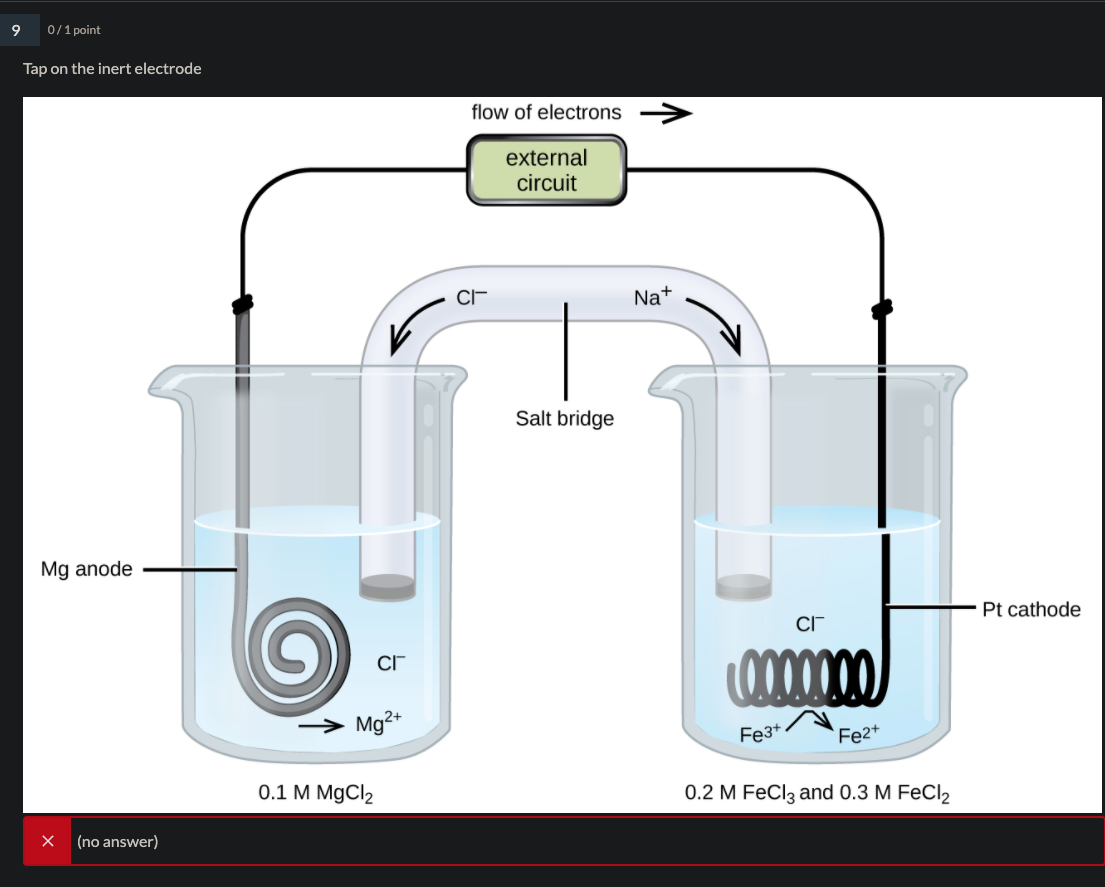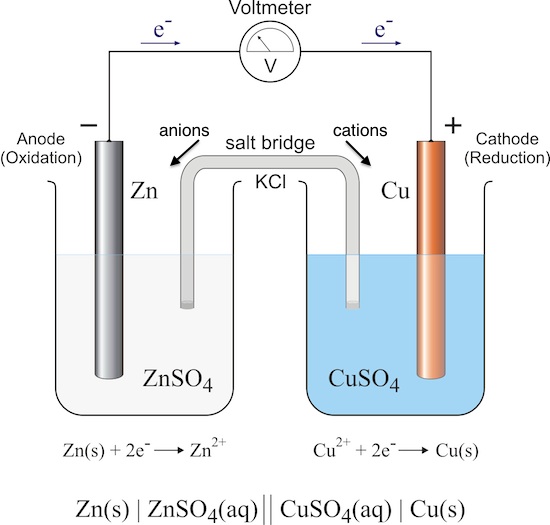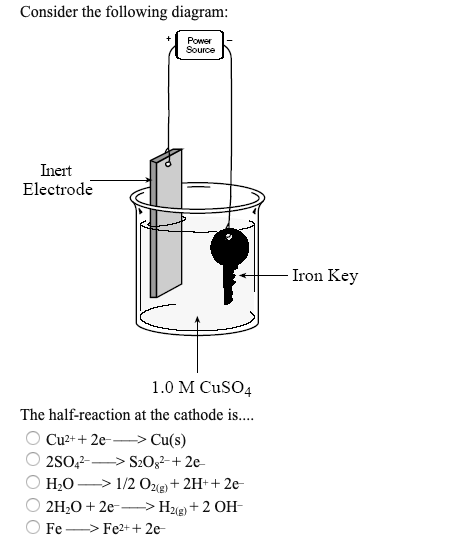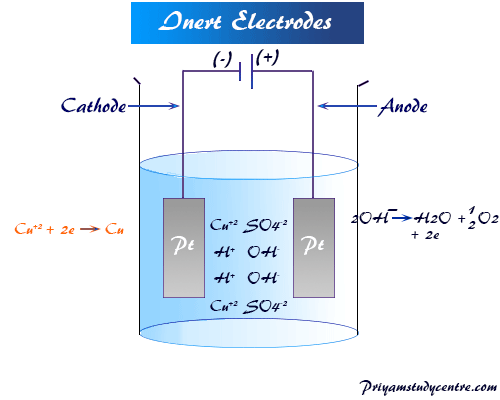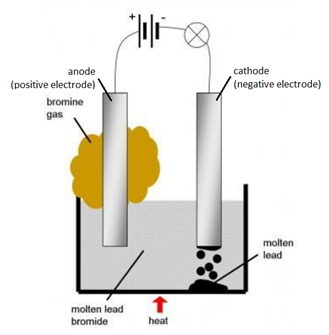
1:58 (Triple only) describe experiments to investigate electrolysis, using inert electrodes, of molten compounds (including lead(II) bromide) and aqueous solutions (including sodium chloride, dilute sulfuric acid and copper(II) sulfate) and to predict