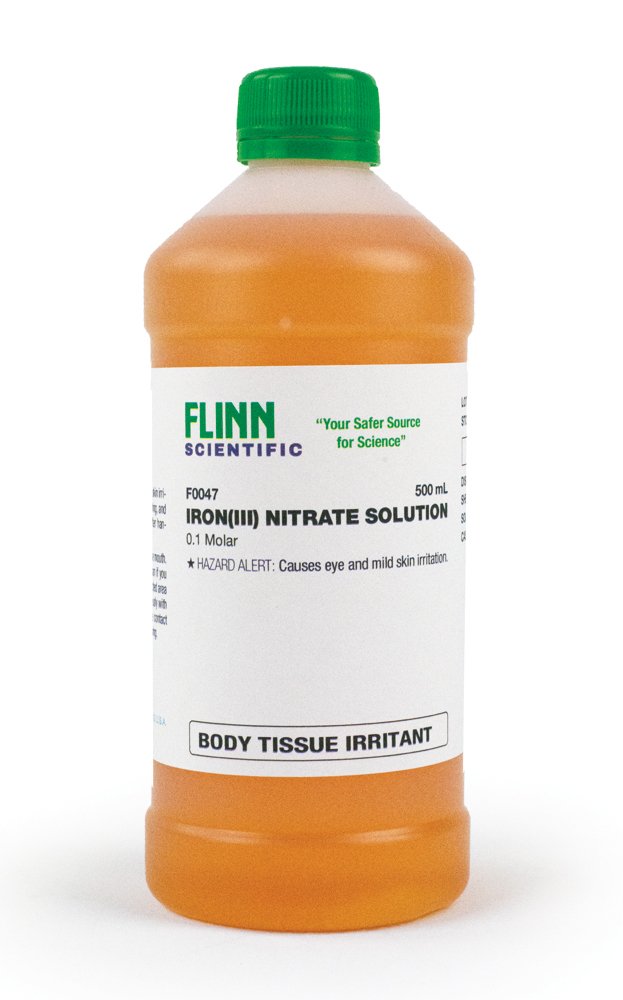
The photographs below (a) show what occurs when a solution of iron(III) nitrate is treated with a few drops of aqueous potassium thiocyanate. The nearly colorless iron(III) ion is converted to a
![7782-61-8・Iron(III) Nitrate Nonahydrate, 99.9%・097-02812・091-02815[Detail Information]|Laboratory Chemicals-FUJIFILM Wako Pure Chemical Corporation 7782-61-8・Iron(III) Nitrate Nonahydrate, 99.9%・097-02812・091-02815[Detail Information]|Laboratory Chemicals-FUJIFILM Wako Pure Chemical Corporation](https://labchem-wako.fujifilm.com/sc/01/7782-61-8.png)
7782-61-8・Iron(III) Nitrate Nonahydrate, 99.9%・097-02812・091-02815[Detail Information]|Laboratory Chemicals-FUJIFILM Wako Pure Chemical Corporation







![iron (III) nitrate] iron (III) nitrate]](https://www.degruyter.com/document/doi/00.0000/IUPAC.iupac.compound.25251/asset/images/25251.png)








![Ferric nitrate (Iron [III] nitrate) – 98% – Nice Chemicals Ferric nitrate (Iron [III] nitrate) – 98% – Nice Chemicals](https://nicechemicals.com/wp-content/uploads/2019/04/Nice-Demo-Pic.png)