A new approach to the equilibrium study of iron(III) thiocyanates which accounts for the kinetic instability of the complexes particularly observable under high thiocyanate concentrations - ScienceDirect
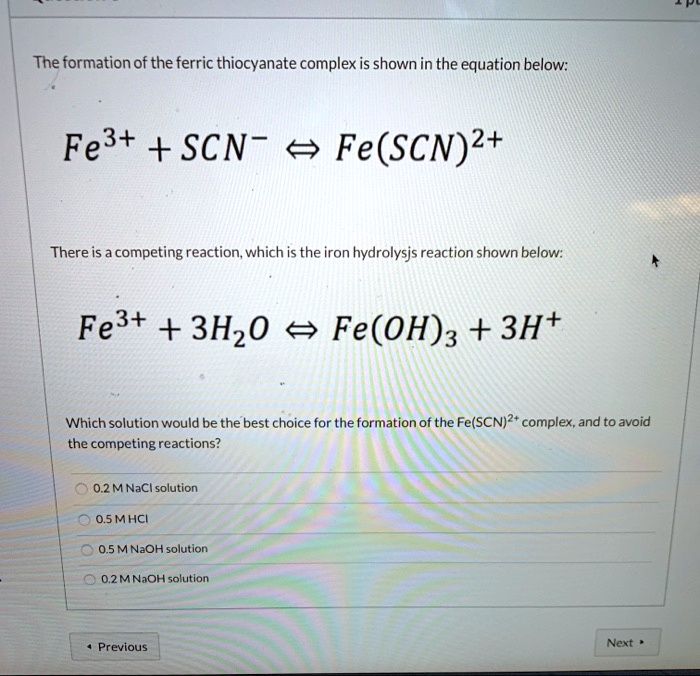
SOLVED: The formation of the ferric thiocyanate complex is shown in the equation below: Fe3+ + SCN- Fe(SCN)+ There is a competing reaction; which is the iron hydrolysjs reaction shown below: Fe3t +

The formation of the iron(III) thiocyanate complex ion is an exothermic equilibrium system. For each change - Brainly.com

Solved: calculate iron(iii) thiocyanate complex ion concentration (in m) for each solution. record in lab - Brainly.com

Reaction scheme showing the formation of iron(III)-thiocyanate complex... | Download Scientific Diagram

SOLVED: The reaction between iron (III) and thiocyanate is an equilibrium reaction as given by the equation below: Fe' - (aq) SCN (aq) FeSCN (aq) Write an equilibrium expression for the iron (
![SOLVED: Concentration iron(IlI) nitrate [Fe(NO3)3] (M) 0.20000 Color of iron (III) nitrate yellow Concentration potassium thiocyanate [KSCN] (M) 0.00200 Color of potassium thiocyanate colorless Volume iron(III) nitrate (mL) 10.00 Volume potassium ... SOLVED: Concentration iron(IlI) nitrate [Fe(NO3)3] (M) 0.20000 Color of iron (III) nitrate yellow Concentration potassium thiocyanate [KSCN] (M) 0.00200 Color of potassium thiocyanate colorless Volume iron(III) nitrate (mL) 10.00 Volume potassium ...](https://cdn.numerade.com/ask_images/14220882a7b444129a38c31a16b7bb26.jpg)