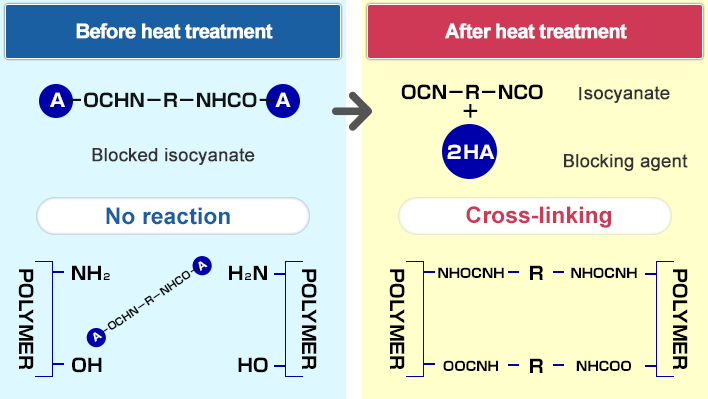
Catalytic Room‐Temperature C−N Coupling of Amides and Isocyanates by Using Mechanochemistry - Dayaker - 2020 - ChemSusChem - Wiley Online Library
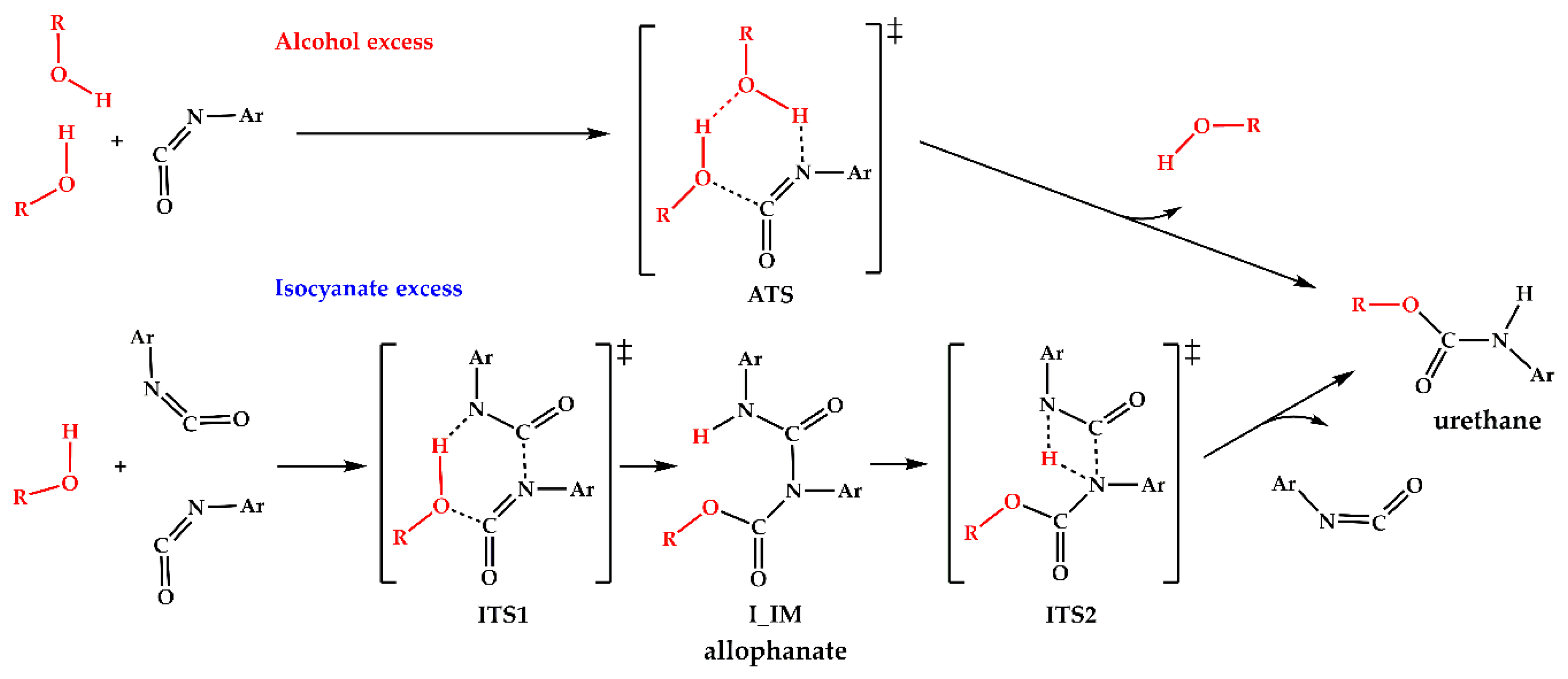
Polymers | Free Full-Text | Urethane Formation with an Excess of Isocyanate or Alcohol: Experimental and Ab Initio Study

Show the mechanism of the nucleophilic addition reaction of an alcohol with an isocyanate to yield a urethane. | Homework.Study.com
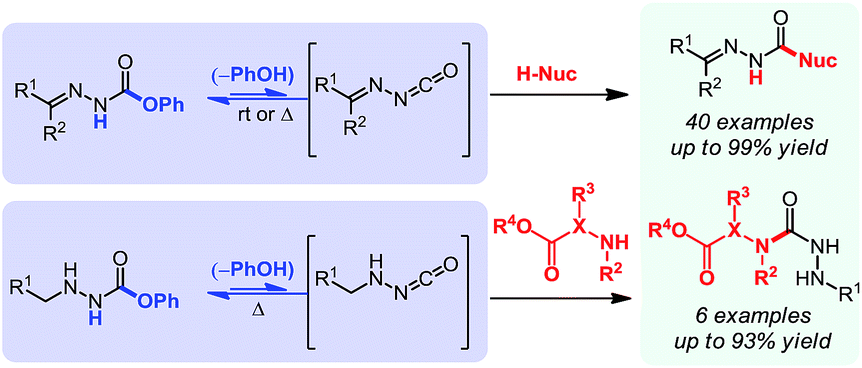
Cascade reactions of nitrogen-substituted isocyanates: a new tool in heterocyclic chemistry - Chemical Science (RSC Publishing) DOI:10.1039/C5SC03197D

A Practical and General Amidation Method from Isocyanates Enabled by Flow Technology - Williams - 2018 - Angewandte Chemie - Wiley Online Library

Thiol−Isocyanate−Ene Ternary Networks by Sequential and Simultaneous Thiol Click Reactions | Chemistry of Materials

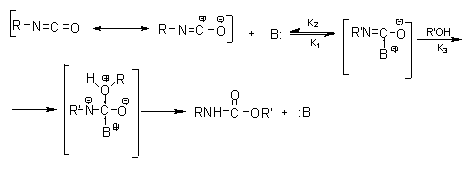
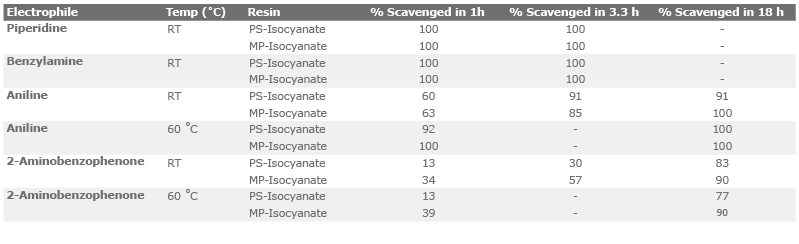


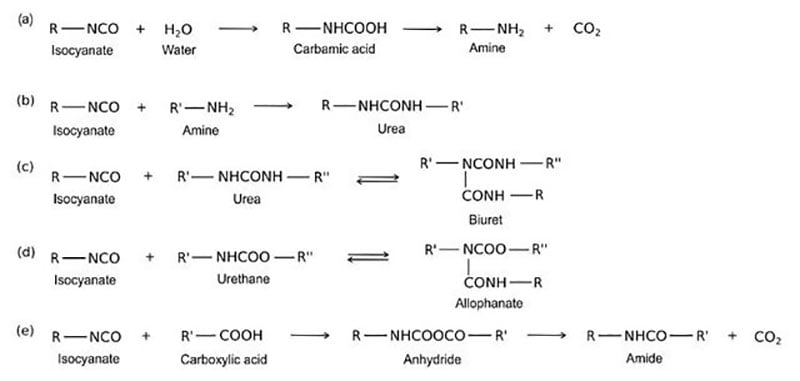
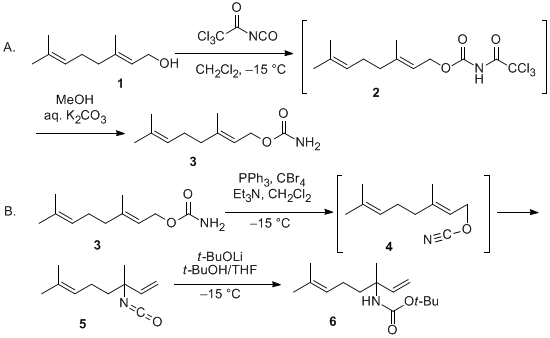



![PDF] A study of elementary reactions of isocyanate production | Semantic Scholar PDF] A study of elementary reactions of isocyanate production | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3d7c2c88b04137199c776d187783553eff2079a3/8-Figure1-1.png)