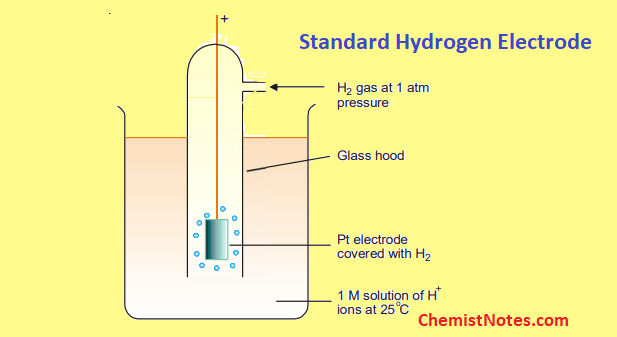
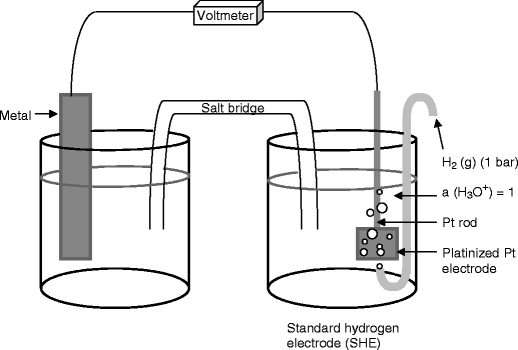
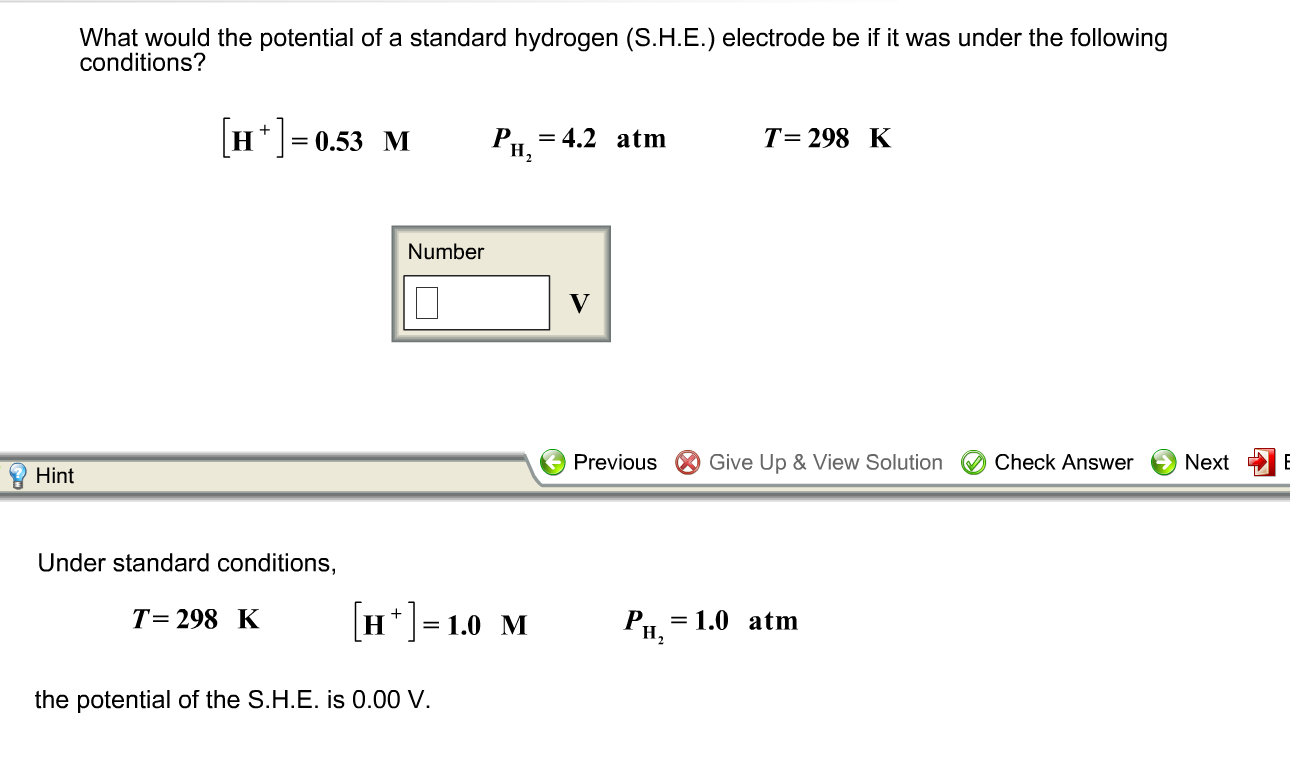
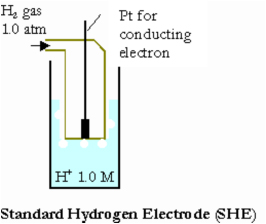
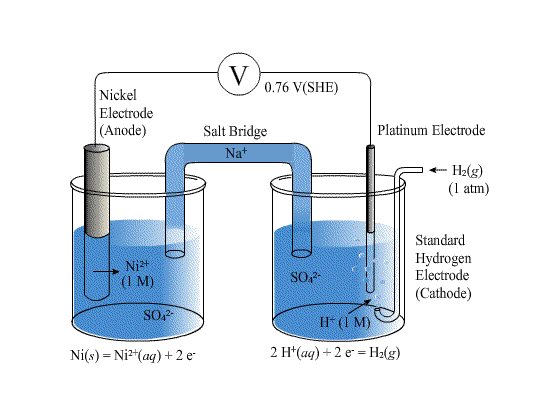
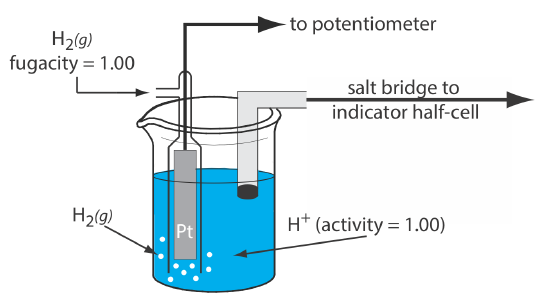
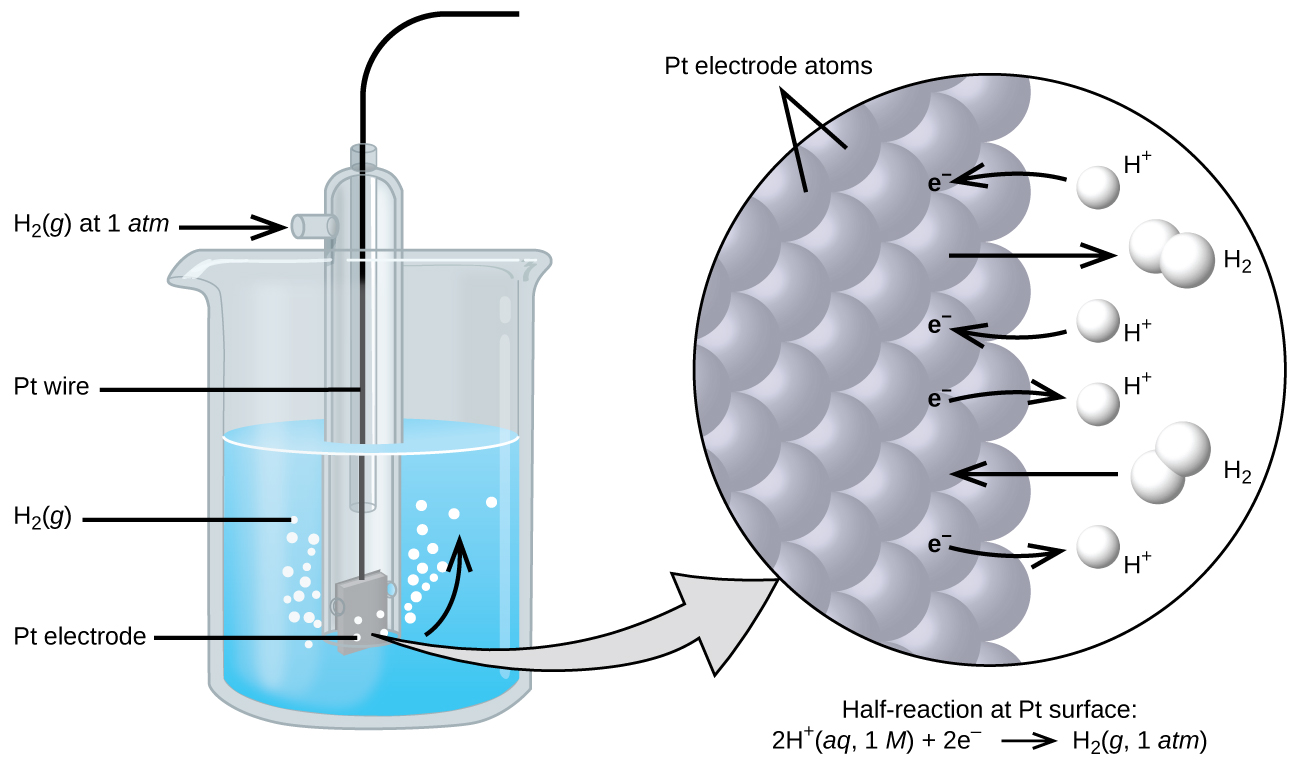
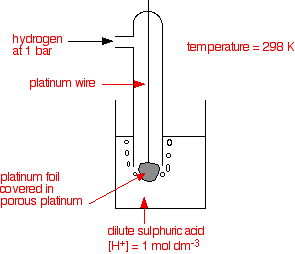
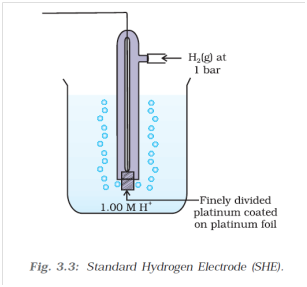
Standard Reference Electrode Standard Hydrogen Electrode (SHE) SHE: Assigned V Can be anode or cathode Pt does not take part in reaction Difficult. - ppt download

Why Standard Hydrogen Electrode act both as a Cathode and Anode | Electrochemistry - Best Online Free Chemistry Learning

Standard and Reversible Hydrogen Electrodes: Theory, Design, Operation, and Applications | ACS Catalysis

Electrode Potentials Standard Hydrogen Electrode Standard Reduction Electrode Chemistry Course Topics Stock Illustration - Download Image Now - iStock