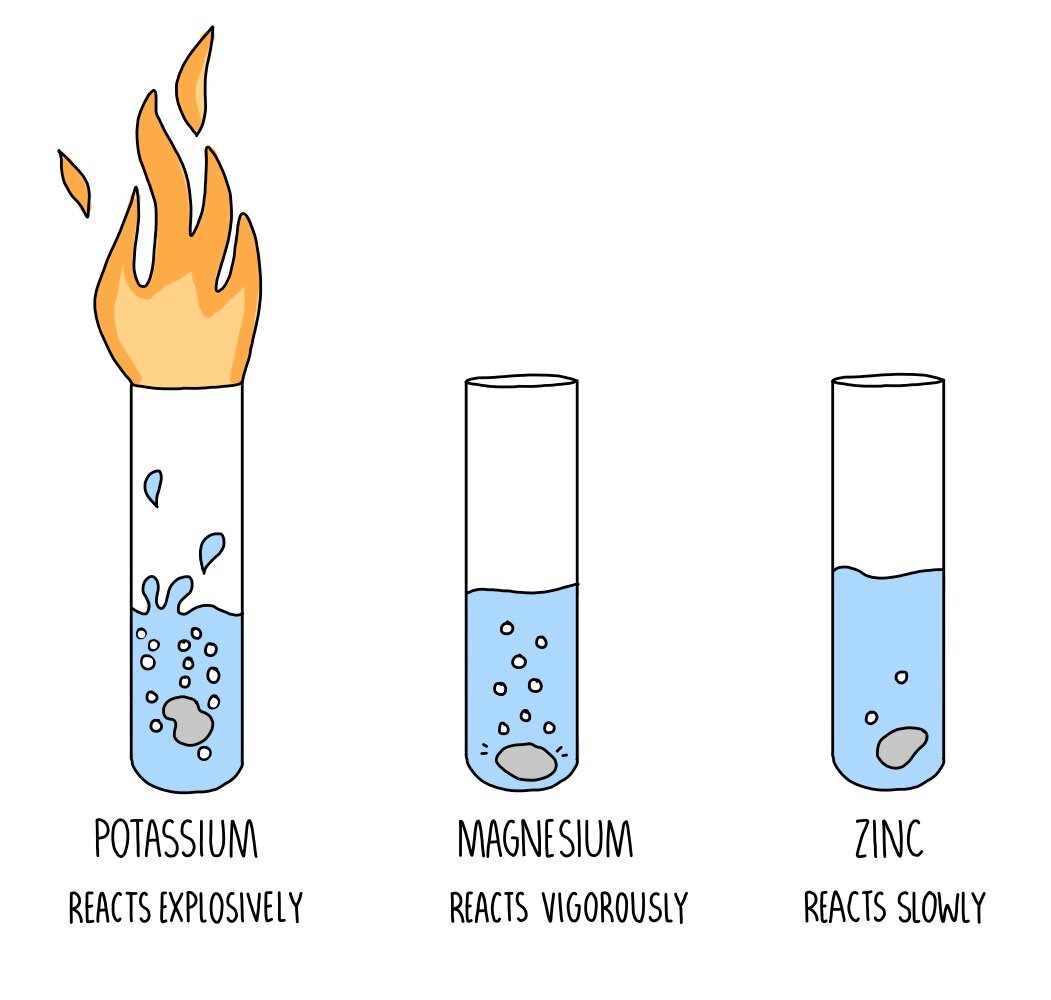
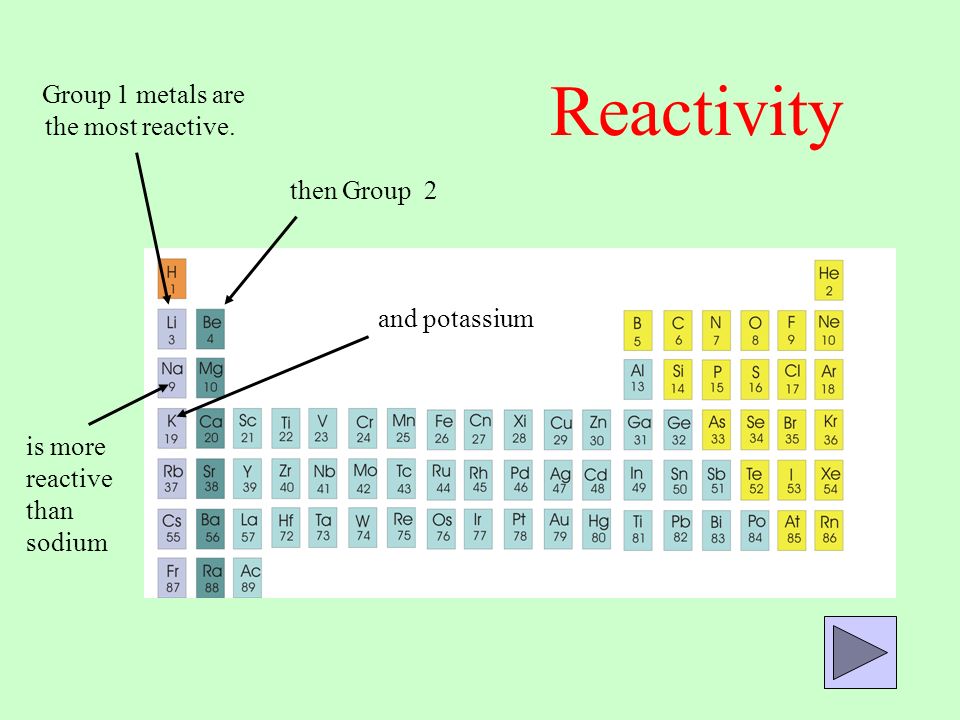
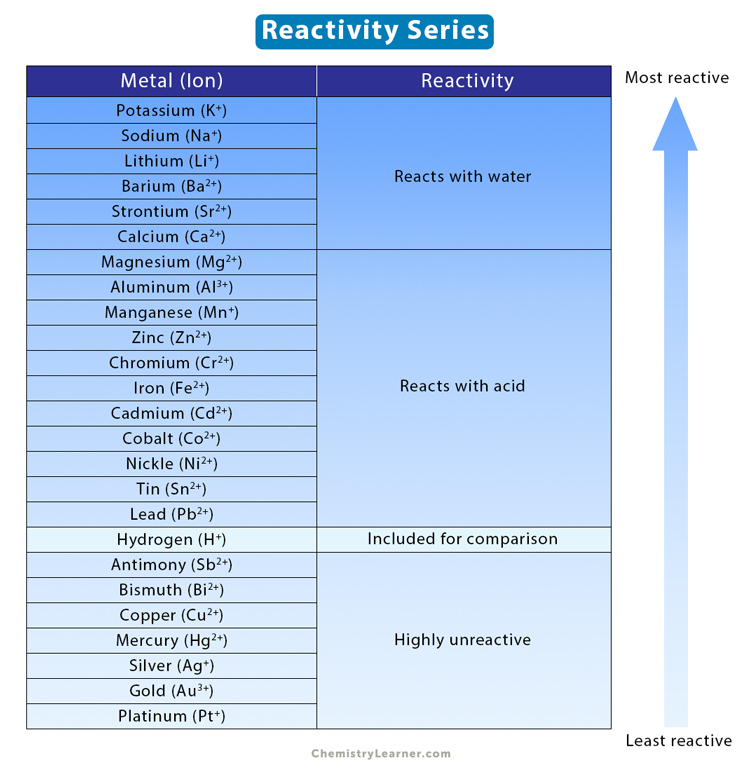
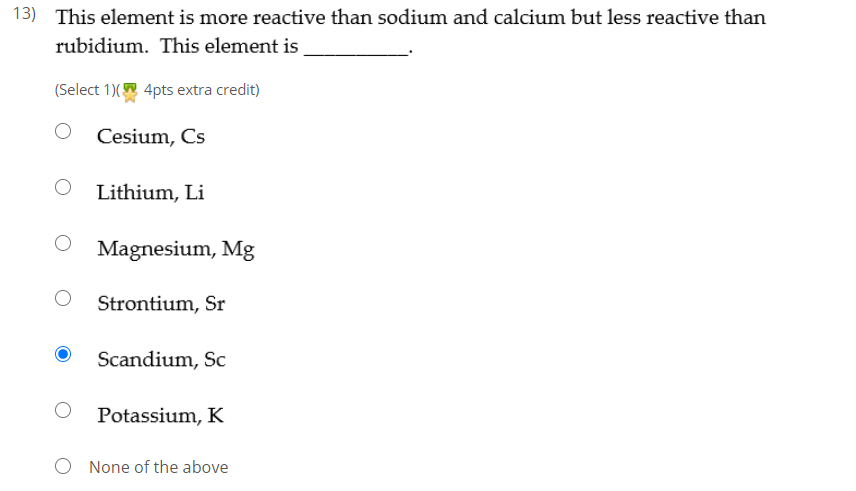15 i) Why is potassium more reactive than lithium ii) Why is fluorine is more reactive than chlorine - Science - - 9764717 | Meritnation.com

In group 1 (alkali metals), sodium is more reactive than lithium, or lithium is less reactive than sodium. Give reason.

a) why is potassium more reactive than lithium b) why fluorine is more reactive tahn chlorine - Chemistry - Periodic Classification of Elements - 12977123 | Meritnation.com